Despite its major role in cellular biology, metabolism has only recently acquired a principal role in the research of the most profound cellular cycle disturbance, cancerous transformation. Myelodysplastic syndromes (MDS), a massively heterogeneous group of Hematopoietic Stem/ Progenitor Cell (HSC/HPC) disorders lie at the interface of normal differentiation and malignant transformation and have thus drew great attention due to their polymorphic presentation and elusive pathophysiology. Failure to establish a direct etiopathogenic relationship with specific genetic aberrations, along with the novel finding of a highly deregulated HIF1 activity by several unrelated research groups worldwide, including ours, urged us to investigate the metabolomic status of human bone marrow derived differentiating myeloid lineage in comparison with one another as well as with control samples.
BM aspiration samples collected from 14 previously untreated MDS patients (10 patients with <5% (1 SLD, 8MLD, 1del5q, group 1- G1) and 4 with >5% BM blasts (2 EB1, 2 EB2group 2 - G2)) and 5 age matched controls. Myeloid lineage cells were isolated through ficoll bilayer protocol. All samples contained homogenous myeloid lineage subpopulations, assessedthrough optical microscopy. Two different metabolite extraction protocols were applied. The one with the best metabolites yield (50% MeOH, 30% ACN, 20% H2O) was chosen. LC-MS/MS analysis was performed using UPLC 1290 system (Agilent Technologies) coupled to a TripleTOF 5600+ mass spectrometer (SCIEX) equipped with SWATH acquisition, SelexION technology and an electrospray ionization source (ESI). A threshold of a minimum of three samples expressing a given metabolite was set against data sparsity. Data tables were scaled by data centering and setting unit variance. Log2 Foldcalculation and PLS analysis were performed for the two datasets (positive and negative ion-modes). R2 and Q2 for positive ion-mode and negative-ion mode analyses were determined. Both datasets were merged in a unique data table by taking into account maximum absolute log2 foldvalues, when a metabolite was found in both datasets.
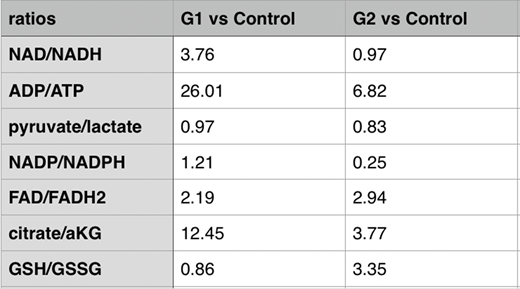
Warburg effect was evidently present in both the G1 and G2 vs control comparisons, yet the role of this stem like aerobic glycolysis seems markedly different in the two groups. While in the G2 group it serves to rescue glucose from complete burn in the mitochondrion and thus shuts it towards nucleotide synthesis (Pentose Phosphate Pathway found upregulated) with the added benefit of increased reduced Glutathione synthesis and improved redox state, in the G1 group proves detrimental. This greatly variable effect of the same phenomenon in the cellular fate lies upon the quality and functionality of the cellular mitochondrial content. G2 precursors presented functional mitochondrial (decreased NAD/NADH and FAD/FADH2) contrary to the G1 ones (Table). Failing TCA cycle, with increased NAD/NADH and FAD/FADH2 ratios and markedly increased ADP/ATP levels leads to FAs accumulation due to failure of effective adequate β oxidation. The uncontrolled increase in the NAD/NADH ratio stimulates upper glycolysis into a turbo mode further increasing the ADP/ATP, depleting cellular energy contents, engaging it to a never-ending deadly metabolism. The enormous abundance of upper glycolytic intermediates is relieved through phospholipid and ceramide synthesis, all found massively upregulated in both the MDS vs control yet also in the G1 vs G2 comparisons. FAs, mostly phospholipid and ceramide accumulation, interrupt the mitochondrial membrane lipidome further incapacitating metabolic integrity and inducing their autophagic degradation which further stimulates the Warburg effect. This type of metabolic reprogramming is eventually targeted to epigenetic modifier production, increased S-adenosyl-methionine, the major methyl group donor, 2-HydroxyGlutarate, a potent epigenetic modifier and notorious oncometabolite, Acetyl-Lysine, the major acetyl- group donor, even glutathione.
We therefore present a model of an uncontrolled Warburg effect which in the G1 group confers premature death of the hematopoietic precursors, the ineffective hematopoiesis of MDS. Yet, under the pressure of the vastly upregulated epigenetic modifiers cellular fate changes, the G1 precursors adapt and transform to the G2 ones yet eventually to Acute Myeloid Leukemia blasts.
Vassilopoulos:Genesis pharma SA: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees; Gilead: Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal